Your cart is currently empty!
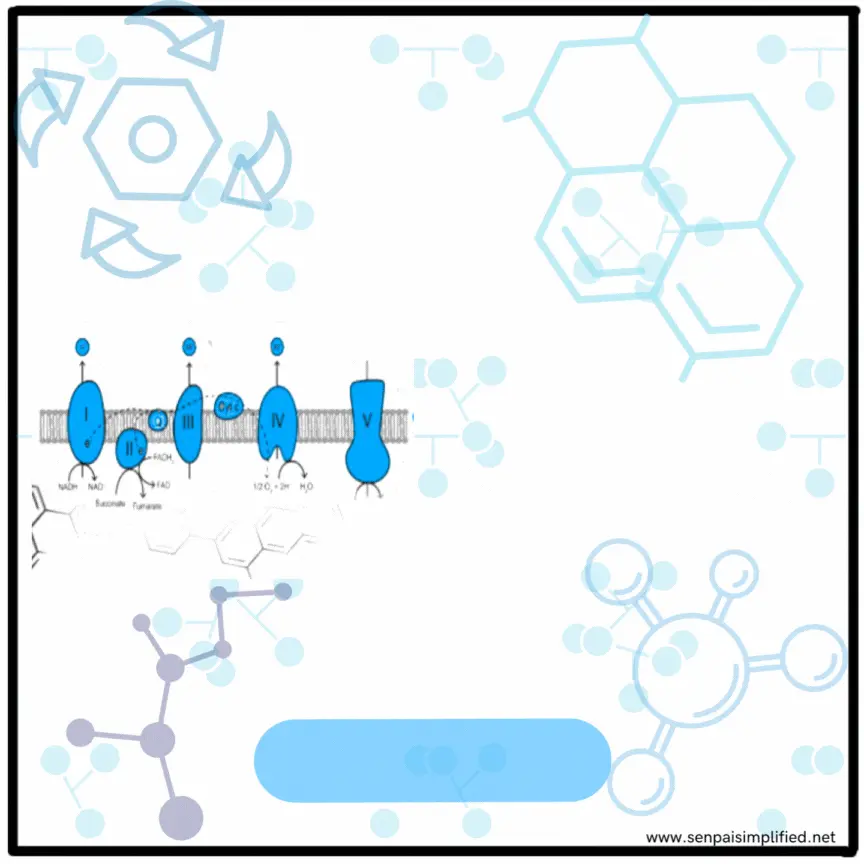
Electron Transport Chain
The electron transport chain (ETC) is a series of protein complexes that are responsible for the production of energy in the form of ATP. Proteins facilitate the transport of ATP, ADP, and Pi in and out of the cell. Additionally, NADH enters through two shuttle systems. One simpler method involves the transporter for malate, bringing NADH from outside the cell to the inside. However, this process is less effective compared to the malate-aspartate shuttle because it results in the production of FADH2.
Mitochondrial Matrix
The mitochondrial matrix is the innermost compartment of the mitochondria, and it contains the enzymes necessary for the TCA cycle, the electron transport chain, and the synthesis of ATP. The outer membrane of the mitochondria is freely permeable to most substances, while the inner membrane is not permeable to all substances. They require special proteins and shunts to transport these molecules across it. Proteins facilitate the transport of ATP, ADP, and Pi in and out of the cell. Additionally, NADH enters through two shuttle systems. One simpler method involves the transporter for malate, which brings NADH from outside the cell to the inside. However, this process is less effective compared to the malate-aspartate shuttle because it results in the production of FADH2.
Complexes I-IV
Complex I
Complex I, also termed NADH-ubiquinone oxidoreductase, initiates the ETC by receiving electrons from NADH and transferring them to the electron carrier Coenzyme Q (CoQ). Consequently, protons are pumped across the inner mitochondrial membrane, creating a proton gradient. This gradient later facilitates ATP generation. Additionally, Complex I contains FMN and Fe-S as coenzymes.
Complex II
In contrast, Complex II, or succinate-ubiquinone oxidoreductase, accepts electrons from FADH2 and transfers them to CoQ. Unlike Complex I, Complex II does not transport protons across the inner mitochondrial membrane, thus no ATP generation occurs. It comprises FAD and Fe-S as coenzymes..
Complex III
Moving on to Complex III, also known as ubiquinol-cytochrome c oxidoreductase, it accepts electrons from CoQ and transfers them to the electron carrier cytochrome c. This process results in the pumping of protons across the inner mitochondrial membrane, establishing a proton gradient. Furthermore, Complex III contains cytochrome bc1 as a coenzyme.
Complex IV
Finally, Complex IV, termed cytochrome c oxidase, receives electrons from cytochrome c and transfers them to oxygen, resulting in the production of water. Simultaneously, it pumps protons across the inner mitochondrial membrane, contributing to the proton gradient. Complex IV includes cytochrome c oxidase as a coenzyme.
Redox Pairs
In the process of oxidation, one substance loses electrons while another gains them, resulting in reduction. This released energy serves as a crucial source for ATP production, vital for cellular energy needs. It’s noteworthy that as electrons move along the chain, from one molecule to another, there’s an increase in the standard reduction potential. For instance, NADH, a potent electron donor, initiates the chain, while oxygen, a robust electron acceptor, concludes it. This intricate interplay of electron transfer not only drives ATP synthesis but also underscores the essential role of each molecule in the chain, from start to finish.
ATP Synthase complex/ Complex V
Energy is stored as the PMF. When H+ re-enters the matrix, it causes rotation of the c ring. Rotation of c ring causes a conformational change in β subunit. ADP is combined with Pi to form ATP (oxidative phosphorylation).
Inhibitors of Electron Transport Chain
Site-specific inhibitors:
| Complex I | Retinone, Amobarbital |
| Complex II | Malonate |
| Complex III | Antimycin A |
| Complex IV | Carbon monoxide and Cyanide [totally arrest respiration] |
| Complex V (ATP synthase) | Oligomycin |
Uncouplers
Typically, the electron transport chain (ETC) and ATP synthesis operate as coupled processes, sharing a common intermediate in the form of the H+ gradient. However, certain proteins form channels that permit H+ to enter the matrix without concurrent ATP synthesis. Instead, the energy released in this process dissipates as heat. This phenomenon is naturally observed in brown adipose tissue, particularly through thermogenin (also known as UCP1). Moreover, synthetic uncouplers, such as 2,4-dinitrophenol (DNP), can mimic this uncoupling effect by disrupting the coupling between ETC and ATP synthesis. These uncouplers increase the permeability of the inner mitochondrial membrane to protons, allowing them to flow back into the matrix without driving ATP synthesis. While this uncoupling phenomenon can increase energy expenditure and has been explored for weight loss purposes, it can also be harmful if not controlled, leading to excessive heat production and potential toxicity.









Leave a Reply